
COVID-19 Diagnostics
NEW SARS-CoV-2 Neutralizing Antibodies Detection Kit
AdipoGen Life Sciences has developed a sensitive, specific and fast SARS-CoV-2 Neutralizing Antibodies Detection Kit (Prod. No. AG-48B-0002) that can be used to detect the presence of neutralizing antibodies against SARS-CoV-2 in serum/plasma in about 2 houts independently of species and isotypes!.
This Detection Kit is based on a colorimetric reaction, which measures the binding of the RBD of the Spike S protein from SARS-CoV-2 to its human receptor ACE2. The presence of neutralizing/blocking antibodies in the samples are detected by reduction of signal indicating the inhibition of the Spike-ACE2 binding.
The SARS-CoV-2 Neutralizing Antibodies Detection Kit contains key reagents required to test the presence of functional neutralizing antibodies against SARS-CoV-2 present in the serum or plasma. It is an easy, fast and scalable (automation) alternative to the classical neutralization assays using Vero E6 cells, such as virus neutralization test (VNT) and pseudo-virus neutralization test (pVNT) and does not need biosafety requirements.
This kit would be administrable in vaccine and therapeutic development as it is suitable for all antibody isotypes and can be used to determine neutralizing antibodies in animal models. The kit will also be useful in current COVID-19 investigations of sero-prevalence, assessment of immune response, herd immunity, longevity of protective immunity, efficacy of different vaccine candidates as well as tracking infections in animals.
NEW & UNIQUE SARS-CoV-2 (Spike RBD) IgG Serological ELISA Kit
AdipoGen Life Sciences has developed a sensitive and specific SARS-CoV-2 (Spike RBD) IgG Serological ELISA Kit (Prod. No. AG-45B-0020) that can be used for the qualitative measurement of human Immunoglobulin G (IgG) against SARS-CoV-2 Spike (Receptor Binding Domain) protein in serum and plasma samples.
During an infection, IgM antibodies appear first, followed by IgA on mucosal surfaces or IgG in the serum. The spike (S) proteins (mainly the RBD domain) and the nucleocapsid (N) proteins are the main immunogens of coronaviruses leading to an antibody answer. This assay is an indirect ELISA assay (based on a coated Spike (RBD) protein).
Included in this assay and unique in the market for SARS-CoV-2 IgG assays, is a second plate (antigen non-coated Background Plate or False Positive Control Plate) to determine the sample specific background and to measure the amount of IgG antibodies non-specifically bound to the well. To measure presence of anti-SARS-CoV-2 human IgG antibodies in serum or plasma, net sample optical densities (Net OD) are calculated by subtracting each sample Background Plate OD from the Spike (SARS-CoV-2) antigen plate (Spike Plate) OD.
Note: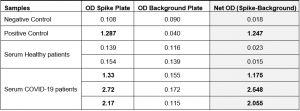
- • In the first week of the onset of the infection with the novel coronavirus (SARS-CoV-2) patients results may be negative for IgG.
- • Patients with low immunity or other diseases that affect immune function, failure of important systemic organs, and use of drugs that suppress immune function can also lead to negative results of new coronavirus IgG.
- • Previous infection of SARS or other coronavirus strains may cause a light IgG positive in view of similarities in the nucleocapsid protein, but not in the Spike RBD protein. This means that nucleocapsid-based SARS-CoV-2 IgG assays have a higher chance of cross-reactivites with other coronavirus IgG antibodies than Spike RBD-based SARS-CoV-2 IgG assays, such as this newly developed assay from AdipoGen Life Sciences.
SARS-CoV-2 Inhibitor Screening Set (Ref. AG-44B-0007) has developed a High Throughput Screening (HTS) detection assay for the determination of SARS-CoV-2 blocking reagents.
The SARS-CoV-2 Inhibitor Screening Kit contains key reagents required to facilitate identification of SARS-CoV-2 inhibitors. This inhibitor screen is based on a colorimetric ELISA kit, which measures the binding of the RBD of the Spike S protein from SARS-CoV-2 to its human receptor ACE2.
NEW ACE2 (human) ELISA Kit
AdipoGen Life Sciences has developed a sensitive and specific colorimeteric sandwich ACE2 (human) ELISA Kit (Prod. No. AG-45B-0023) that can to be used for the in vitro quantitative determination of soluble human ACE2 in cell culture supernatants, serum, plasma and urine.
 ACE2 has been identified as a key receptor on target cells for SARS-CoV infections in 2002. ACE2 functions as the entry receptor of the new SARS-CoV-2 coronavirus that emerged in China in 2019 and is the cause of the new disease COVID-19. Strong binding of the spike protein of SARS-CoV-2 to ACE2, along with proteolytic cleavage of ACE2 by transmembrane serine protease 2 (TMPRSS2), facilitates entry of the virus into cells, viral replication and cell-to-cell transmission. ACE2 can undergo an ADAM17 (a disintegrin and metalloproteinase 17)-mediated “shedding” from endothelial cells, resulting in the release of the ectodomain into the circulation. This soluble form may act as a competitive interceptor of SARS-CoV-2 and other coronaviruses by preventing binding of the viral particle to the surface-bound, full-length ACE2. A reliable ELISA Kit to determine soluble ACE2 in the circulation can be useful in therapeutic developmental studies.
ACE2 has been identified as a key receptor on target cells for SARS-CoV infections in 2002. ACE2 functions as the entry receptor of the new SARS-CoV-2 coronavirus that emerged in China in 2019 and is the cause of the new disease COVID-19. Strong binding of the spike protein of SARS-CoV-2 to ACE2, along with proteolytic cleavage of ACE2 by transmembrane serine protease 2 (TMPRSS2), facilitates entry of the virus into cells, viral replication and cell-to-cell transmission. ACE2 can undergo an ADAM17 (a disintegrin and metalloproteinase 17)-mediated “shedding” from endothelial cells, resulting in the release of the ectodomain into the circulation. This soluble form may act as a competitive interceptor of SARS-CoV-2 and other coronaviruses by preventing binding of the viral particle to the surface-bound, full-length ACE2. A reliable ELISA Kit to determine soluble ACE2 in the circulation can be useful in therapeutic developmental studies.
Soluble ACE2 might also be used as biomarker of hypertension and cardiovascular diseases.
Validated Recombinant Proteins for ACE2 & COVID-19 Research
| Product Name | Cat. No. | Source | Purity | Application / Activity |
| ACE2 (human) (rec.) | AG-40B-0192 | HEK293 cells | ≥95% (SDS-PAGE) | Soluble human ACE2 competitively inhibits SARS-CoV-2 infection. |
| NEW ACE2 (human) (rec.) (Biotin) | AG-40B-0192B | HEK293 cells | ≥95% (SDS-PAGE) | Soluble human ACE2 competitively inhibits SARS-CoV-2 infection. |
| NEW ACE2 (human) (rec.) (His) | CHI-B232008 | HEK293 cells | ≥90% (SDS-PAGE) | Soluble human ACE2 competitively inhibits SARS-CoV-2 infection. |
| ACE2 (human):Fc (human) (rec.) | CHI-B232006 | HEK293 cells | ≥90% (SDS-PAGE) | Soluble human ACE2 competitively inhibits SARS-CoV-2 infection. |
| ACE2 (mouse) (rec.) | AG-40B-0193 | HEK293 cells | ≥95% (SDS-PAGE) | Soluble mouse ACE2 negative control. |
| SARS-CoV-2 Spike Protein S1 (RBD) (rec.) (His) | CHI-B232004 | HEK 293 cells | ≥90% (SDS-PAGE) | Soluble Protein S (RBD) competitively inhibits SARS-CoV-2 infection. For drug and antibody screening applications and immunization. |
| NEW SARS-CoV-2 Spike Protein S1 (RBD) (rec.) (GST-His) | CHI-B249001 | HEK 293 cells | ≥95% (SDS-PAGE) | Soluble Protein S (RBD) competitively inhibits SARS-CoV-2 infection. For drug and antibody screening applications and immunization. |
| SARS-CoV-2 Spike Protein S1 (RBD):Fc (human) (rec.) | CHI-B232003 | HEK 293 cells | ≥95% (SDS-PAGE) | Soluble Protein S (RBD) competitively inhibits SARS-CoV-2 infection. For drug and antibody screening applications and immunization. |
| SARS-CoV-2 Spike Protein S1 (RBD):Fc (human) (rec.) | AG-40B-0194 | HEK 293 cells | ≥95% (SDS-PAGE) | Soluble Protein S (RBD) competitively inhibits SARS-CoV-2 infection. For drug and antibody screening applications and immunization. |
| SARS-CoV-2 Nucleocapsid Protein (rec.) (His) | CHI-B233501 | E. coli | ≥95% (SDS-PAGE) | For drug screening applications. |
| PLpro (SARS Coronavirus) (rec.) (His) | SBB-DE0024 | E. coli | ≥95% (SDS-PAGE) | Involved in the processing of the viral polyprotein. |
| ISG15 (human) (rec.) (Rhodamine 110) | SBB-PS0002 | E. coli | ≥97% (LCMS) | PLPro substrate. Inhibits viral budding and acts as IFNγ-inducing cytokine. |
UNIQUE Human ACE2 Monoclonal Blocking Antibody
AdipoGen Life Sciences’ anti-ACE2 (human), mAb (blocking) (AC384) (preservative free) (Prod. No. AG-20A-0037PF) is a monoclonal antibody that recognizes human ACE2 and works specifically in ELISA, Western Blot and Functional Application. The antibody blocks the binding of human ACE2 to the Spike protein of SARS-Cov-2.
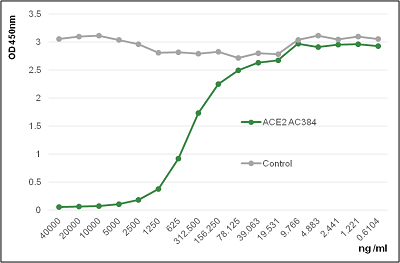
Figure: Binding of ACE2 (human) to the Spike protein of SARS-CoV-2 is inhibited by the antibody anti-ACE2 (human), mAb (blocking) (AC384) (AG-20A-0037PF).
Method: SARS-CoV-2 Spike Protein S1 (RBD):Fc (human) (rec.) (AG-40B-0194) is coated on an ELISA plate at 1µg/ml. ACE2 (human), mAb (blocking) (AC384) (AG-20A-0037PF) or an unrelated mAb Control are added (starting at 40µg/ml with a two-fold serial dilution) together with 500ng/µl of ACE2 (human) (AG-40B-0192). After incubation for 1h at RT, the binding was detected using an anti-FLAG antibody (HRP).
Flow Cytometry-Competent Human ACE2 Monoclonal Antibodies
AdipoGen Life Sciences’ anti-ACE2 (human), mAb (AC18F) (Prod. No. AG-20A-0032) is a monoclonal antibody clone that recognizes human ACE2 and works specifically in Flow Cytometry (FACS). The antibody is available in different formats, unlabeled (#AG-20A-0032) and Biotin-labeled (#AG-20A-0032B), as well as labeled with the dyes ATTO488 (#AG-20A-0032TD) and ATTO647N (#AG-20A-0032TS). All variants are FACS-competent using the appropriate secondary reagents.
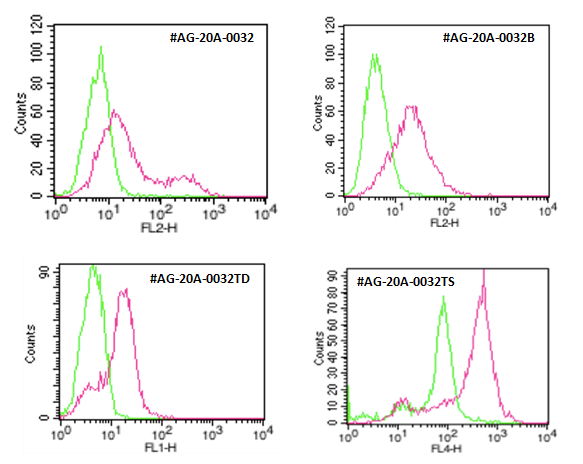
Figure: Detection of endogenous human ACE2 by different formats of anti-ACE2 (human), mAb (AC18F) (AG-20B-0032).
Method: HepG2 cell line is stained with anti-ACE2 (human), mAb (AC18F) (red line) or an appropriate isotype control at 1µg/106 cells each, revealed with a secondary reagent and then analyzed by flow cytometry.
Download: Cell Detachment Protocol for FACS.
| Product Name | Cat. No. | Isotype | Application | Species |
| anti-ACE2 (human), mAb (AC18F) | AG-20A-0032 | Mouse IgG1κ | ELISA, FACS, WB | Human |
| anti-ACE2 (human), mAb (AC18F) (Biotin) | AG-20A-0032B | Mouse IgG1κ | ELISA, FACS, WB | Human |
| anti-ACE2 (human), mAb (AC18F) (ATTO 488) | AG-20A-0032TD | Mouse IgG1κ | FACS | Human |
| anti-ACE2 (human), mAb (AC18F) (ATTO 647N) | AG-20A-0032TS | Mouse IgG1κ | FACS | Human |
| anti-ACE2 (human), mAb (AC384) | AG-20A-0037 | Mouse IgG1κ | ELISA, WB | Human |
| anti-ACE2 (human), mAb (AC384) (Biotin) | AG-20A-0037B | Mouse IgG1κ | ELISA, WB | Human |
| anti-ACE2 (human), mAb (blocking) (AC384) (preservative free) | AG-20A-0037PF | Mouse IgG1κ | FUNC (Blocking), ELISA, WB | Human |
| anti-ACE2 (human), pAb | AG-25A-0042 | Rabbit | ELISA, WB | Human |
Antiviral Compounds – Potential Small Molecule Therapeutics Against COVID-19
There are no approved drugs to treat the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection that causes coronavirus disease 2019 (COVID-19). Existing drugs, that have a known favorable safety profile, are being examined for strategies to treat the disease and fast-track a treatment plan. Several influenza and HIV drugs are currently undergoing clinical trial in coronavirus patients. The rational selection of drugs already on the market is being made based on their ability to inhibit any proteins essential for virus-receptor interaction and/or viral life cycle.
AdipoGen Life Sciences offers a Selection of Antiviral Small Molecules as Potential Tools for in vitro Studies of COVID-19 (not for human use).
| Product Name | Cat. No. | CAS Number | Target | Antiviral activity |
| NEW Aloxistatin [E-64d] | AG-CR1-3737 | 88321-09-9 | Viral entry Cathepsin L |
Viral Replication |
| Amastatin . hydrochloride | AG-CP3-7003 | 100938-10-1 | Viral entry ANPEP (Aminopeptidase N) |
Viral Replication |
| NEW Baricitinib | AG-CR1-3734 | 1187594-09-7 | JAK1/JAK2 / Immunomodulation AP-2 associated protein kinase 1 / Endocytosis |
Viral Replication |
| NEW Boceprevir | AG-CR1-3735 | 394730-60-0 | Main Protease (Mpro) | Viral Transcription/Replication |
| Camostat mesylate | AG-CR1-3716 | 59721-29-8 | Viral entry TMPRSS2 |
Viral Replication |
| Chloroquine . diphosphate | AG-CR1-3721 | 50-63-5 | Lysosome function Zinc ionophore Immunomodulation |
Viral Replication |
| Hydroxychloroquine . sulfate | AG-CR1-3720 | 747-36-4 | Lysosome function Zinc ionophore Immunomodulation |
Viral Replication |
| NEW Clevudine | AG-CR1-3736 | 163252-36-6 | RNA-dependent RNA polymerases (RdRps) | Viral Transcription/Replication |
| Darunavir | AG-CR1-3712 | 206361-99-1 | Papain-like viral protease (PLVP) | Viral Maturation/Replication |
| Darunavir . ethanolate | AG-CR1-3724 | 635728-49-3 | Papain-like viral protease (PLVP) | Viral Maturation/Replication |
| NEW Dexamethasone | AG-CR1-3742 | 50-02-2 | Inflammatory Factors | Cytokine Storm |
| Ebselen | AG-CR1-0031 | 60940-34-3 | Main Protease (Mpro)/3C-like Protease | Viral Transcription/Replication |
| NEW EIDD-2801 | AG-CR1-3733 | 2349386-89-4 | RNA-dependent RNA polymerases (RdRps) | Viral Transcription/Replication |
| Elbasvir | AG-CR1-3729 | 1370468-36-2 | RdRP, Papain-like Proteinase and Helicase | Viral Replication |
| NEW Famotidine | AG-CR1-3730 | 76824-35-6 | SARS-CoV-2 papain-like protease (PLpro) | Viral Replication |
| Favipiravir | AG-CR1-3717 | 259793-96-9 | RNA-dependent RNA polymerases (RdRps) | Viral Transcription/Replication |
| NEW Ibudilast | AG-CR1-3738 | 50847-11-5 | Macrophage Inhibitory Factor (MIF) Immunomodulation |
Viral Replication Cytokine Storm |
| Imatinib mesylate |
AG-CR1-3725 | 220127-57-1 | Virion fusion with endosomal membrane | Viral Replication |
| Lopinavir | AG-CR1-3715 | 192725-17-0 | Coronavirus endopeptidase C30 (CEP_C30) | Viral Maturation/Replication |
| Mycophenolic acid |
AG-CN2-0419 | 24280-93-1 | SARS-CoV-2 papain-like protease (PLpro) | Viral Replication |
| NEW Nafamostat mesylate | AG-CR1-3731 | 82956-11-4 | Viral entry TMPRSS2 |
Viral Replication |
| Nelfinavir . mesylate |
AG-CR1-3726 | 159989-65-8 | Post-entry inhibitor | Viral Replication |
| Niclosamide |
AG-CR1-3643 | 50-65-7 | Endosome acidification | Viral Replication |
| Niclosamide . ethanolamine |
AG-CR1-3644 | 1420-04-8 | Endosome acidification | Viral Replication |
| Nitazoxanide |
AG-CR1-3723 | 55981-09-4 | Viral hemagglutinin Viral IE2 |
Viral Maturation/ Transcription/Replication |
| Oseltamivir . phosphate |
AG-CR1-3714 | 204255-11-8 | Viral neuraminidase Release of viral particles |
Viral Replication |
| NEW PX-12 | AG-CR1-3739 | 141400-58-0 | Main Protease (Mpro)/3C-like Protease | Viral Transcription/Replication |
| Remdesivir |
AG-CR1-3713 | 1809249-37-3 | RNA-dependent RNA polymerases (RdRps) | Viral Transcription/Replication |
| Remdesivir Metabolite GS-441524 |
AG-CR1-3722 | 1191237-69-0 | RNA-dependent RNA polymerases (RdRps) | Viral Transcription/Replication |
| Ribavirin |
AG-CR1-3719 | 36791-04-5 | RNA-dependent RNA polymerases (RdRps) RNA capping activity Viral mutation rates Immunomodulation |
Viral Transcription/Replication |
| Ritonavir |
AG-CR1-3683 | 155213-67-5 | Coronavirus endopeptidase C30 (CEP_C30) | Viral Maturation/Replication |
| Rosuvastatin . calcium salt |
AG-CR1-3728 | 147098-20-2 | Main Protease Mpro | Viral Replication |
| Ruxolitinib . phosphate salt |
AG-CR1-3645 | 1092939-17-7 | JAK1/JAK2 Immunomodulation |
Viral Replication |
| Ruxolitinib (free base) |
AG-CR1-3624 | 941678-49-5 | JAK1/JAK2 Immunomodulation |
Viral Replication |
| Saquinavir . mesylate |
AG-CR1-3727 | 149845-06-7 | Main Protease Mpro | Viral Replication |
| Shikonin |
AG-CN2-0487 | 517-89-5 | Main Protease (Mpro)/3C-like Protease | Viral Transcription/Replication |
| NEW Suramin . hexasodium salt | AG-CR1-3575 | 129-46-4 | Viral Entry/Fusion RNA-dependent RNA polymerases (RdRps) |
Viral Transcription/Replication |
| NEW TDZD-8 | AG-CR1-3740 | 327036-89-5 | Main Protease (Mpro)/3C-like Protease | Viral Transcription/Replication |
| NEW Telaprevir | AG-CR1-3741 | 402957-28-2 | 3C-like Protease | Viral Replication |
| Tofacitinib |
AG-CR1-3625 | 477600-75-2 | JAK1/JAK3 Immunomodulation |
Viral Replication |
| Tofacitinib citrate |
CDX-T0461 | 540737-29-9 | JAK1/JAK3 Immunomodulation |
Viral Replication |
| Umifenovir . HCl [Arbidol] |
AG-CR1-3718 | 131707-23-8 | Viral entry Fusion into host cells |
Viral Replication |
Cytokine Storm and Severity of COVID-19
The most critically ill COVID-19 patients are known to undergo a cytokine storm leading to poor prognosis and need of urgent anti-inflammatory treatment/hospitalization. There are many variations on this phenomenon and they go by many names: systemic inflammatory response syndrome, macrophage activation syndrome or cytokine release syndrome (CRS).
Therefore the diagnostic detection and treatment of cytokine storms has become an important part of rescuing severe patients. Targets like IL-1, IL-6, IL-7, IL-10, IL-18, IL-33, IFN-γ, TNF-α or many others might play an important role in cytokine release syndrome (CRS). Detection of their levels and blockage of their signaling pathways with immunomodulatory agents (biologicals, small molecules) is expected to become a new method for the treatment of severe patients.
AdipoGen Life Sciences offers a broad range of Cytokine Immunoassays as well as recombinant cytokines and blocking antibodies, which are already being successfully used to research the various cytokine storm factors involved and to characterize the immune response.

