Muchas pruebas de diagnóstico para la enfermedad por COVID-19 están disponibles hasta el momento. En esta entrada de blog, nos vamos a centrar en mostrar las características y las principales ventajas para ser utilizados en la detección, el cribado y la monitorización de la enfermedad.
Introducción
Se está desarrollando una gama de técnicas moleculares que van desde pruebas de laboratorio para el diagnóstico y el manejo de pacientes con COVID-19 hasta pruebas pruebas que puedes hacer en casa. Estas técnicas, aunque son bien conocidas por investigadores y médicos, así como los ensayos de anticuerpos y antígenos, pueden ser relativamente desconocidas para la comunidad en general. Esta entrada de blog se ha escrito para describir y explicar los principios básicos de las principales pruebas de diagnóstico de COVID-19 actualmente en uso.
Transcripción inversa y reacción en cadena de la polimerasa (RT-PCR)
La PCR es una técnica muy común que se ha sido (y es) utilizada en investigación y diagnóstico durante los últimos 30 años para detectar información genética. La RT-PCR es una versión de esta técnica para detectar la presencia de ARN. Actualmente, esta técnica se está utilizando como prueba para detectar la presencia del virus SARS-CoV-2. Este tipo de prueba se ha utilizado con frecuencia como prueba de primera línea para COVID-19, ya que prueba directamente la presencia del ARN del virus.
Las pruebas de RT-PCR son bastante rápidas, sensibles y confiables, capaces de producir resultados en 3-4 horas, aunque esto generalmente toma más tiempo si las muestras deben enviarse primero a laboratorios externos especializados (6-8 horas en promedio). Muchas compañías de diagnóstico e investigación producen productos, pruebas y máquinas de RT-PCR para que la tecnología esté ampliamente disponible. Algunas pruebas de RT-PCR se desarrollan como un kit «todo en uno», lo que reduce la manipulación en el laboratorio y el potencial de contaminación.
¿Cómo funciona?
Una vez que se recoge la muestra, se deben eliminar proteínas y otras moléculas, aislando tan sólo el ARN. Esta será una mezcla del material genético de una persona, así como cualquier ARN viral que pueda estar presente.

Las enzimas del kit transcriben el ARN en el ADN, que se amplifica para permitir la detección de virus mediante el uso de un termociclador que genera un programa de temperaturas para producir aproximadamente 35 mil millones de copias de ADN viral para cada cadena de ARN viral que estuvo originalmente presente. Se utilizan marcadores fluorescentes que son capaces de unirse al ADN amplificado y producir luz, que la máquina puede leer para producir el resultado de la prueba.
Si la intensidad de la luz producida alcanza un cierto umbral, esta se clasifica con un resultado positivo. Se registra el número de ciclos de temperatura de PCR que se requerían antes de alcanzar el umbral de fluorescencia y proporciona una estimación de la cantidad de virus presente en la muestra del paciente. En primer lugar, el ADN viral se calienta a 94ºC, separando las dos hebras del ADN. Acto seguido, la reacción se enfría a 55ºC, Este hecho, permite que pequeñas porciones de ADN complementario (primers o cebadores) se unan al ADN viral.

Finalmente, se eleva la temperatura a 72ºC para que la Taq polimerasa pueda elongar cada una de estas hebras de ADN. Y de esta manera, se inicia un nuevo ciclo de PCR.
¿Qué detecta el test de RT-PCR?

La RT-PCR detecta si el ARN viral está presente o no en las muestras de un paciente. Lo hace amplificando regiones del material genético del virus, generalmente la proteína Spike, la proteína N o la envoltura del virus. El resultado de la RT-PCR indica el número de ciclos necesario para detectar un umbral de detección del ADN, de tal forma que a más virus presente en la muestra, menor número de ciclos necesarios para obtener la señal. Comúnmente, estas muestras se toman de la nariz o la garganta usando hisopos largos o cortos.
¿Qué significa el resultado obtenido?
Una prueba de RT-PCR es altamente sensible y su resultado aporta información sobre si el virus está presente en el paciente.
- Resultado positivo de la prueba: Un resultado positivo de la PCR significa que la persona de la que se tomó la muestra está actualmente infectada por el virus.
- Resultado negativo de la prueba: Un resultado negativo de PCR podría significar que: la persona no está infectada actualmente por este virus, o que el virus no está presente en el lugar donde se tomó la muestra, también un falso negativo podría ser fruto de una muestra de baja calidad o que es demasiado pronto o demasiado tarde en la infección para detectar la replicación del virus. Esta es la razón por la cual los resultados negativos de la prueba requieren que se tomen nuevas muestras de pacientes unos días después para reducir la posibilidad de extrañar incorrectamente a una persona infectada.
La prueba RT-PCR no puede detectar si una persona ha tenido el virus y superó la enfermedad COVID-19, ya que solo detecta cuando hay virus activo.
Ventajas:
- RT-PCR es una técnica robusta.
- Dado que la RT-PCR es tan común en los laboratorios, la tecnología ya está en funcionamiento para detectar la presencia del virus.
- RT-PCR puede detectar infecciones en curso, lo que permite al personal médico determinar quién está infectado actualmente y quién no.
Limitaciones:
- RT-PCR se basa en la detección del virus, por lo que a efectos de recuento, es posible pasar por alto aquellos pacientes que han eliminado el virus y se han recuperado de la enfermedad.
- La distribución del virus a través del tracto respiratorio varía entre los pacientes, por lo que incluso si una persona está infectada, el virus a veces es solo detectable en el esputo o en el hisopo nasofaríngeo, pero no necesariamente en ambos lugares al mismo tiempo.
- La RT-PCR para COVID-19 solo puede determinar si una persona está actualmente infectada con este coronavirus en particular. No puede proporcionar información sobre otras enfermedades o síntomas.
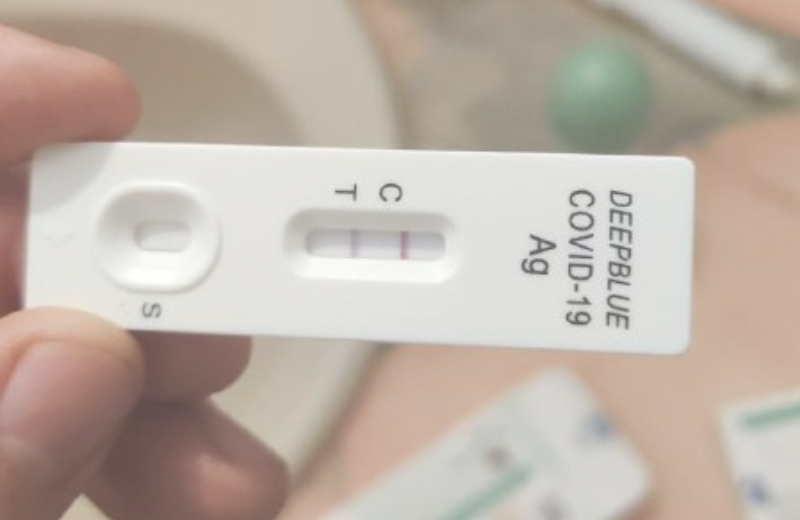
Flujo Lateral / Inmunocromatografía coloidal

Los ensayos de flujo lateral inmunocromatográfico se han denominado comúnmente «Test rápidos» en los medios de comunicación, ya que actualmente se utilizan para detectar anticuerpos contra la enfermedad en la sangre de un paciente. Las pruebas de flujo lateral pueden detectar anticuerpos contra el virus en la sangre del paciente, lo que indica que el paciente tiene COVID-19 o se ha recuperado de COVID-19.
Las pruebas de flujo lateral de anticuerpos para SARS-CoV-2 deben ser prescritas y utilizadas por un personal sanitario. Requieren una gota de sangre del paciente, normalmente procedente de un pinchazo en un dedo, similar a las pruebas para controlar el azúcar en la sangre en ciertos tipos de diabetes. Estos tests funcionan de manera muy diferente a la técnica de RT-PCR y se centran en detectar la respuesta inmunológica del paciente al virus en lugar de detectar el virus en sí.
¿Cómo funciona?
Los inmunoensayos de flujo lateral son dispositivos simples que pueden detectar anticuerpos en la sangre. Personal sanitario toma una pequeña muestra de sangre del paciente y la deja caer sobre una esponja dentro del dispositivo. A continuación, se agregan unas gotas de tampón que ayudan a que la muestra de sangre fluya a través del dispositivo. A medida que la muestra se mueve a través del dispositivo, los anticuerpos contra el SARS-CoV-2 que están presentes en la muestra se capturarán los anticuerpos en las líneas de prueba y control. Este proceso de captura y unión produce un cambio de color a lo largo de las líneas de prueba y control que puede verse a simple vista, produciendo una, dos o tres líneas dependiendo del tipo de anticuerpos presentes (IgM o IgG).

¿Qué detecta el test de flujo lateral?
Los inmunoensayos de flujo lateral para SARS-CoV-2 detectan dos tipos de anticuerpos protectores que son producidos por el cuerpo cuando el sistema inmunitario se activa. Estos anticuerpos ayudan a combatir la enfermedad y permanecen en la sangre durante meses después de que el virus y la enfermedad se eliminan. La presencia de anticuerpos en el cuerpo a menudo se denomina inmunidad o que una persona es inmune a un virus, ya que estos anticuerpos protegen contra la reinfección y el retorno de la misma enfermedad.
Cuando estamos infectados por un virus, nuestro sistema inmunitario produce anticuerpos tempranos del tipo IgM con una unión de fuerza intermedia al virus, que pueden comenzar a trabajar para eliminar el virus aproximadamente 5 días después de una nueva infección. Por lo general, de 8 a 10 días después de la infección, los anticuerpos IgG con alta fuerza de unión pueden ayudar a eliminar el virus más rápidamente. Los inmunoensayos de flujo lateral de anticuerpos pueden diseñarse para detectar IgM o IgG solos o ambos juntos.
¿Qué significa el resultado obtenido?
Los inmunoensayos de flujo lateral de anticuerpos detectan anticuerpos contra el virus en la sangre. No detectan el virus en sí. Las pruebas de anticuerpos proporcionan una capacidad enormemente importante para detectar infecciones pasadas con virus para identificar a las personas que fueron asintomáticas, las personas que eliminaron el virus y, por lo tanto, que ya no corren el riesgo de infectarse o transmitir el virus a otras personas. Además, las pruebas de anticuerpos son críticas para evaluar la propagación del virus a la población y el nivel de inmunidad del «rebaño» en la población. Esto es importante para comprender las posibles consecuencias de levantar o aplicar medidas para controlar el virus, como la cuarentena, el distanciamiento social, el cierre de escuelas y los lugares de trabajo.

Las pruebas de inmunoensayo de flujo lateral para evaluar la presencia de anticuerpos IgG e IgM son muy simples de interpretar: Debe aparecer una línea de control para mostrar que el ensayo ha funcionado correctamente. Otros resultados que pueden aparecer son alguno de los tipos de anticuerpos se encuentra en la muestra. La aparición de líneas para IgG o IgM, o ambas, indica una muestra positiva y por lo tanto, que el paciente ha sido infectado con el coronavirus COVID-19.
Ventajas:
- Los ensayos de flujo lateral son extremadamente rápidos, aportan resultados en solo 15 minutos.
- Permiten el muestreo no invasivo para detectar la presencia del virus.
Limitaciones:
- La precisión en el diagnóstico de coronavirus es limitada.
- Hasta ahora, los kits de flujo lateral disponibles solo pueden determinar si un paciente ha sido infectado en algún momento con COVID-19. Se necesitarían más pruebas para verificar si un paciente está actualmente infectado.
Inmunoensayos tipo ELISA
Un ELISA es una técnica común que se puede utilizar para detectar antígenos o anticuerpos. Los ensayos ELISA utilizan enzimas unidas a anticuerpos que causan un cambio de color que puede ser cuantificado. La fuerza del cambio de color es proporcional al número de anticuerpos presentes en la muestra. Los ELISA permiten realizar hasta 96 ensayos completados al mismo tiempo, lo que permite disponer de un método económico y efectivo para analizar lotes de con muchas muestras al mismo tiempo. Esta tecnología podría ayudar a acelerar el número de pacientes que se pueden analizar para el SARS-CoV-2.
¿Como funciona?
Un ELISA detecta anticuerpos producidos en la sangre del paciente debido a la infección con SARS-CoV-2. Todo el experimento se puede realizar en un pocillo e implica mezclar muestras de pacientes, anticuerpos, antígenos y enzimas junto con un sustrato que al ser metabolizado se convierte en un producto que cambia de color. Veamos el siguiente diagrama:

- Una muestra de plasma del paciente se añade al pocillo que contiene el antígeno específico de SARS-CoV-2.
- Los anticuerpos presentes (o no) contra el SARS-CoV-2 en el plasma del paciente se unen a los antígenos dispuestos en la base del pocillo, el resto de los elementos presenten en la muestra del plasma son eliminados mediante lavados.
- Se añaden anticuerpos secundarios unidos con enzimas se unen a los anticuerpos del paciente. El exceso de anticuerpo no unido al anticuerpo del paciente es eliminado mediante lavados.
- Se añade el sustrato que es reconocido por las enzimas que están unidas al anticuerpo secundario.
- En los pocillos que se encuentran los anticuerpos contra SARS-CoV-2 empiezan a cambiar de color.
- El producto formado es capaz de ser determinado objetivamente con un lector de placas.
- Si el paciente no fue infectado con COVID-19, todo el proceso no puede ser llevado a cabo, de tal forma que no se forma un cambio de color en el pocillo.
¿Qué detecta el test de ELISA?
Los kits de ELISA para detectar anticuerpos están detectando la respuesta de los anticuerpos a la infección por COVID-19. La detección de anticuerpos contra el SARS-CoV-2 podría decirle a un especialista clínico si un paciente ha sido infectado con COVID-19, ya sea actualmente o en el pasado. Sin embargo, los pacientes infectados no serán detectados inmediatamente después de la infección, sino solo cuando el sistema inmune al virus pueda detectarse en la sangre, aproximadamente 5 días después de la infección. El conocimiento actual sugiere que una vez que una persona ha sido infectada con el virus, su sistema inmunológico evitará una futura infección con el mismo virus.
El kit de ELISA para la presencia de anticuerpos proporciona información muy importante para el diagnóstico, el manejo y la recuperación de COVID-19 y también ayuda a los investigadores a evaluar cuántas personas de la población han sido infectadas, lo cual es importante para planificar el control de infecciones.
¿Qué significa el resultado obtenido?
Un kit de ELISA muestra un resultado positivo (cambio de color del pocillo) si el paciente tiene anticuerpos contra SARS-CoV-2. Esto podría no significar que actualmente tienen el virus, solo que lo han tenido en algún momento. Esto se debe a que los anticuerpos permanecen en la sangre incluso después de que la infección desaparezca. Un resultado negativo significaría que el paciente no ha sido infectado con COVID-19 y puede no tener inmunidad contra él.
Ventajas:
- Los ELISA es una técnica de laboratorio simple y barata.
- Los ELISA son ensayos bien establecidos y documentados dentro de la comunidad científica y médica.
- Los resultados generalmente pueden ser generados de 1 a 3 horas de la recogida de la muestra del paciente.
- Los kits de ELISA permiten evaluar a muchos pacientes a la vez.
Limitaciones:
- No pueden ser realizados in situ, las muestras deben recogerse, ser procesadas y analizadas en un laboratorio.
Conclusiones
- Se están utilizando o desarrollando tres tipos principales de pruebas para detectar el SARS-CoV-2.
- Estas pruebas diagnósticas se encuentran en diferentes etapas de desarrollo, validación y producción.
- Cada tipo de ensayo tiene sus propias ventajas y desventajas inherentes a la tecnología subyacente.
- Una combinación de tipos de prueba utilizados en diferentes momentos puede ser útil para el manejo del paciente y el control de la pandemia de la población.
| Tecnología | Molécula testada | Requisitos | Tiempo necesario | Tipo de muestra | Número de muestras |
| RT-PCR | ARN viral | Laboratorio | 3-4 horas | hisopo nasofaríngeo o esputo | Hasta 96 muestras |
| Flujo Lateral | Anticuerpos IgG y IgM contra SARS-CoV-2 | Punto de asistencia | 15-20 minutos | Sangre | 1 muestra por ensayo |
| ELISA | Anticuerpos IgG y IgM contra SARS-CoV-2 | Laboratorio | 1-3 horas | Sangre | Hasta 96 ensayos |
Si necesitas mas información o tienes cualquier consulta, no dudes en preguntarnos:





Un comentario en “Tipos de tests para detectar el COVID-19”
İnat Box MOD APK
I am happy to find this article content very helpful and valuable for everyone, as it contains lot of topic information.