Western blotting (WB) remains the most commonly used method for separating and detecting proteins, and yet it can be very difficult to make it work to your advantage. Optimizing your protocol is crucial, but, factors such as antibody quality, blocker buffers, and transfer efficiency play key roles.
Let’s take a closer look at the optimizing tips and tools that are available today.
- 1. Load the right amount of protein
- 2. Do my samples need to be reduced or denatured?
- 3. How to avoid the dreaded “smiley” bands and the spots?
- 4. Is there any difference between blocking the membrane with BSA or milk?
- 5. Which antibody dilution should I use?
- Check all the solutions for Western blot available in the SERVA catalog
1. Load the right amount of protein
The quantity of protein that must be loaded in each well is variable and must be optimized by the researcher since it is a factor that depends on the expression levels of that gene in that tissue. In addition, it must be taken into account what volume of sample that well admits. However, between 10 and 50 ug of protein from tissue homogenate and between 10 and 100 ng of total protein in purified samples is usually sufficient.
2. Do my samples need to be reduced or denatured?
To review the technical datasheet of the antibody is essential to know how we should prepare our samples. Usually, samples for WB are usually denatured proteins. The native proteins have a tertiary structure and its conformation confers a fundamental role in the fucntion of the protein. However, this structure will affect the migration of these proteins through the polyacrilamine pores. Protein denaturation can be done by:
- Sodium dodecyl sulfate (SDS), is a detergent that is used to break hydrogen bonds and fall off the tertiary structure.
- Reducing agents such as b-mercaptoethanol or dithiothreitol (DTT) to reduce disulfide bonds between cysteines.
- Heat the samples to 100ºC to denature and facilitate the union of SDS.
3. How to avoid the dreaded “smiley” bands and the spots?
If our membrane has these “smiling bands”, probably electrophoresis must have been carried out too quickly or the samples have become too hot. It is recommended to perform a slower migration for longer and, if possible, cold.
Sometimes, white spots may appear on our blot as a result of air bubbles that have been retained during the transfer process. To avoid this annoying problem, we recommend using a 50 ml conical tube on the transfer set with the idea of eliminating residual air between the polyacrylamide gel and the nitrocellulose membrane or PVDF.
4. Is there any difference between blocking the membrane with BSA or milk?

After transferring the protein from the polyacrylamide gel to the membrane it is essential to block those points of the membrane that do not contain ligand in order to reduce the background or non-specific binding of the antibody with the membrane.
For us, it is more advisable to follow the advice that the manufacturer specifies since usually the antibodies can be very sensitive to the type of blocking agent that is used.
In general, blocking with bovine serum albumin (BSA) provides clearer results since this protein has a lower cross-reactivity. In addition, the use of BSA is indicated in those blots in which we want to detect phosphorylated proteins.
However, many antibodies work better with skimmed milk because it contains a wide variety of blocking agents.
5. Which antibody dilution should I use?
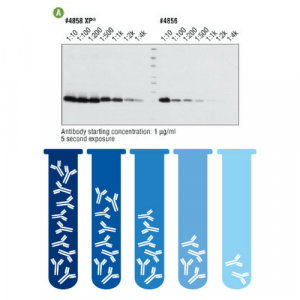 The optimal concentration of an antibody is that by which we obtain a good signal with minimal background noise. Obviously, to save, it is ideal to dilute our antibody as long as the signal is adequate.
The optimal concentration of an antibody is that by which we obtain a good signal with minimal background noise. Obviously, to save, it is ideal to dilute our antibody as long as the signal is adequate.
However, with new antibodies, it is appropriate to make a bank of dilutions in order to know which is the dilution that best adapts to that protein in this tissue. For example, if the manufacturer suggests a 1:200 dilution, we can test dilutions 1:50, 1:100, 1:500 or even 1:1000.
In the case that we do not have additional information about the dilution that we should use, we can orient ourselves according to these parameters:
- supernatant from cell culture: approximately 1:10
- ascites: 1:100
- total antiserum: from 1:50 to 1:100
- purified antibody: 5 ug/mL
Check all the solutions for Western blot available in Labclinics catalog
Genetex offers a selection of products related to Western Blot (electrophoresis, transfer, blocking, etc.).
¿Tienes dudas?
Si no te queda claro del todo cómo funciona esta tecnología, o quieres que te ayudemos a configurar tu ensayo, nuestro departamento técnico de especialistas, con amplia trayectoria en investigación (todos PhD), te pueden echar una mano: por mail (tecnic@labclinics.com), por tlf +34.934464700 o de forma presencial. Contáctanos y estaremos encantados de poder ayudarte!





Leave a reply